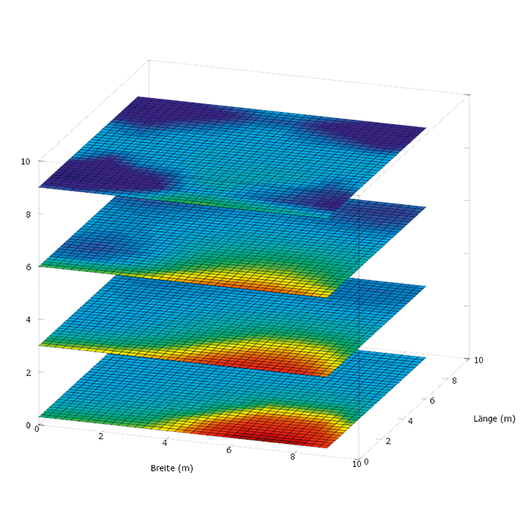
Mapping
Neuberger’s Mapping is the ideal tool for calculating the humidity and temperature distribution in both refrigerated and frozen areas, and in warehouses. Mapping involves taking precise measurements according to requirements such as summer and winter operation and/or loaded and unloaded states in order to be able to precisely determine temperature distributions. The measurement results are displayed using modern 3D graphics. The calibration of the sensors and measuring instruments, the performance of the measurements and the documentation are carried out in compliance with current GMP regulations.
Calibration
Whether you require a comparative measurement, 1- or 3-point calibration, initial calibration or recalibration, Neuberger’s trained engineers perform calibration on-site or in our Rothenburg factory in line with your specifications, and always using certified and tested measuring equipment. The complete measuring section (loop) is calibrated. Of course, these calibrations can also be performed independently. Calibrations are carried out exclusively using reference measuring equipment with Deutsche Akkreditierungsstelle (DAkkS) certification or equivalent. This gives you, the customer, the advantage of traceability.
Our calibration services at a glance:
- Calibrations performed on schedule
- Use of measuring equipment with DAkkS certification or equivalent
- Creation of measuring point lists
- Clear labelling of probes after calibration
- Certificate creation
- Calibration performed by qualified engineers
Validation and qualification
It is not possible to achieve GMP compliance through software and hardware alone. A compliant solution requires the validation, management and organisation of the systems on the basis of corresponding procedural instructions (SOPs). Neuberger can supply you with trusted systems and procedures that have been audited successfully by pharmaceutical companies many times over. We prepare all the requisite documentation, working in conjunction with the customer. Neuberger offers complete software and hardware solutions, consultancy, validation and training, so you only need one contact person for complete GMP monitoring.