Measuring and control technology and monitoring.
From your clean room expert.
As a specialist in building automation, Neuberger is also a leader in the planning, project engineering and installation of measurement, control and regulation equipment for clean rooms. We develop our GMP monitoring systems, airlock controls and clean room terminals entirely in-house and provide complementary services such as mapping, plant qualification and calibration. The main benefit of Neuberger’s clean room technology is the integration of our building automation, monitoring, airlock control and operating control products in clean rooms across multiple buildings.
Innovative system solutions.
Creating a clean environment.
In the clean room technology sector, we are the number one partner for the pharmaceutical industry, life science, medicine, semiconductor and electronics industry, plastics production, laboratories, cosmetics industry and aerospace technology – in short, wherever the maintenance and monitoring of a clean production environment is essential for product quality. In addition to our primary specialism of controlling and conditioning the clean room climate, we offer a comprehensive portfolio of clean room technology. It goes without saying that our products and solutions comply with all national and international guidelines, in particular FDA 21 CFR Part 11 and EU GMP guidelines.
The key feature of our end-to-end GMP monitoring system is the tamper-resistant ProGrafNT software that comes with the PMC2 automation system. Customer benefits: Instrumentation and control technology and clean room monitoring come from a single source. This eliminates measurement errors and investment in duplicate instrumentation. Users can operate and create a visual display of clean room conditions from the outside at any time using our innovative clean room terminals, which can be configured to suit individual requirements. Another key area of our clean room products is the control of personnel and material airlocks.
Neuberger’s unique selling point: We develop and manufacture clean room terminals, airlock controls and programmable logic controllers with an unlimited supply of spare parts, and we offer unlimited updates for ProGrafNT, our building management system.
Evaluations
and reports
In addition to controlling and monitoring stable ambient conditions using programmable logic controllers and ProGrafNT monitoring software, Neuberger also facilitates the evaluation and processing of quality-relevant data, which is another important tool for achieving GMP compliance in clean rooms. With just a few clicks, you can generate audit trails, batch records and well-structured reports relating to limit value monitoring, fault messages, production environments and other critical data. Needless to say, all documentation and reports comply with international standards.
Data logging
and data integrity
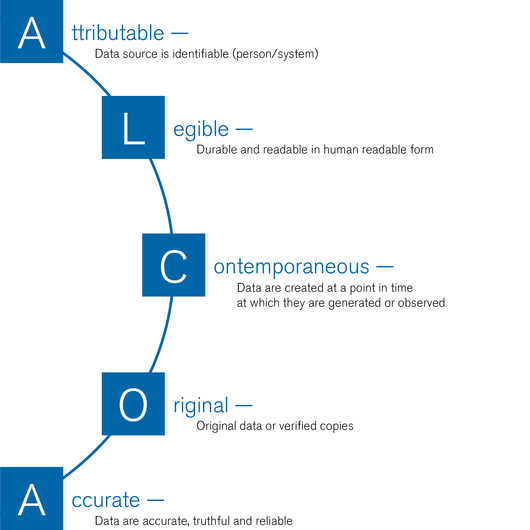
The very essence of clean room and GMP monitoring is the documentation of data throughout the entire development and manufacturing process. In recent years, one buzzword has increasingly come into focus: data integrity. Regulatory bodies have tightened the requirements for the IT security of computerised systems in guidelines such as FDA 21 CFR Part 11, EU GMP Guideline Annex 11, etc. In this environment, spreadsheets have definitely had their day as a reliable form of documentation. Neuberger’s GMP monitoring system provides a tamper-resistant database structure, GMP-compliant recording of all sensitive raw data such as measured values and user interventions, while ensuring data integrity in all circumstances. Our IT security concept follows the ALCOA principle. During GMP audits, you can rely on the fact that the quality-relevant process data will be available in a classifiable, readable, complete and correct format exactly when you need them.

Services
all about clean room technology
A clean room is a complex structure that is permanently subject to equally complex quality controls. This is why regular inspection, qualification and validation of the measurement and plant equipment is also part of our service. Even the slightest deviation in temperature, relative humidity, particle concentration or pressure can have a devastating impact on product quality and safety. Neuberger provides the following clean room technology services: Mapping for the validation of warehouses and climate chambers, GMP-compliant qualification and calibration of measuring points with certified reference measuring equipment. You can also draw on the support of our own nationwide after-sales service team.
Individual solutions
Analysis, planning and project engineering – a single point of contact during all project phases
Development
In-house development of hardware and software, ongoing product development
Manufacturing and production
In-house manufacturing of switchboards and electronic assemblies
Quality assurance
In-house test department – 100% testing of all switchboards. Quality and environmental management according to DIN ISO standards
Installation and commissioning
By more than 170 qualified and regularly trained specialists
Training
In-house training courses, external and on-site customer training, regular seminars, own training center
Full service
Maintenance, 24/7 service, comprehensive service network, personal support through qualified staff
Full product support
Unlimited spare parts, 24/7 service, maintenance